
Luis Batista, PhD
Assistant Professor
Division of Hematology, Department of Medicine
- Email: LBATISTA@nospam.WUSTL.EDU
Awards:
- Pew Foundation Fellow
- California Institute of Regenerative Medicine Scholar
- V Foundation Scholar
- Edward Marllinckrodt Jr Foundation Fellow
Core Usage & WUSTL Affiliations
- Siteman Cancer Center
- Children’s Discovery Institute
- Digestive Disease Research Core Centes
- Genome Editing and iPSC Core (GEiC)
Modeling Human Telomere Disease in Stem Cells
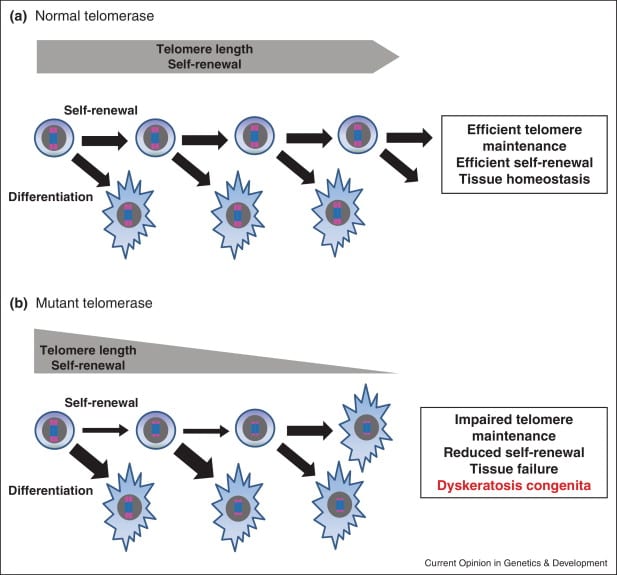
The overarching goal of research in the Batista Lab is to understand the importance of telomerase–the enzyme that adds protective telomore caps to the ends of chromosomes–in human stem and progenitor cell function and regulation.Defects in telomerase, and subsequent telomore shorting, are responsible for a wide range of human disorders, including pulmonary fibrosis, acute myeloid leukemia, and liver cirrhosis. In particular, they are interested in understanding how genetic instability leads to diminished pluripotent cell function over time and how it inuences tissue homeostasis, human agingand cancer. They use genome-wide methods to uncover alterations that drive cellular failure upon critical telomere dysfunction, using human pluripotent cells as a primary model.
Selected Publications
- Fok WC, Niero ELO, Dege C, Brenner KA, Sturgeon CM, Batista LFZ. (2017). p53 Mediates Failure of Human Definitive Hematopoiesis in Dyskeratosis Congenita. Stem Cell Reports. 9:409-418.
- Fok WC and Batista LFZ. Stressed ends: telomere attrition in chronic diseases. (2017). Rev Bras Hematol Hemoter. 39:98-99.
- Batista LF and Artandi SE et al. (2011). Telomore shortening and loss of self-renewal in dyskeratosis congenita induced pluripotent stem cells. Nature 474:399-402.